SareptAlly

SareptAlly is a global program designed to help patients, families, and physicians gain access to resources and information about Sarepta clinical trials and potential treatment options. SareptAlly is dedicated to helping physicians and families in two important ways:
- Sarepta clinical trial identification and matching, or
- assessing potential treatment options
SareptAlly supports individuals with rare diseases in which Sarepta has therapeutic expertise and a physician has determined a patient has an unmet medical need.
Clinical Trial Identification
Well-designed clinical trials are the best way to determine the safety and effectiveness of investigational therapies and for patients to gain access prior to regulatory approval. SareptAlly connects patients with research physicians conducting Sarepta-sponsored clinical research. If a trial is identified, patients may be assessed for trial eligibility by the research physician based on their clinical status and the study protocol.
Local Treatment Assessment
When a clinical trial is not identified, SareptAlly serves as a resource for physicians, patients, and families seeking potential treatment options. SareptAlly responds to inquiries, walks through steps in the process for potentially securing treatment, and identifies resources to help make the process easier.
Other Questions?
For questions not related to clinical trial identification or local access options, Sarepta Patient Affairs team is available at [email protected].
What to Expect When Connecting with SareptAlly: Clinical Trial Identification and Matching
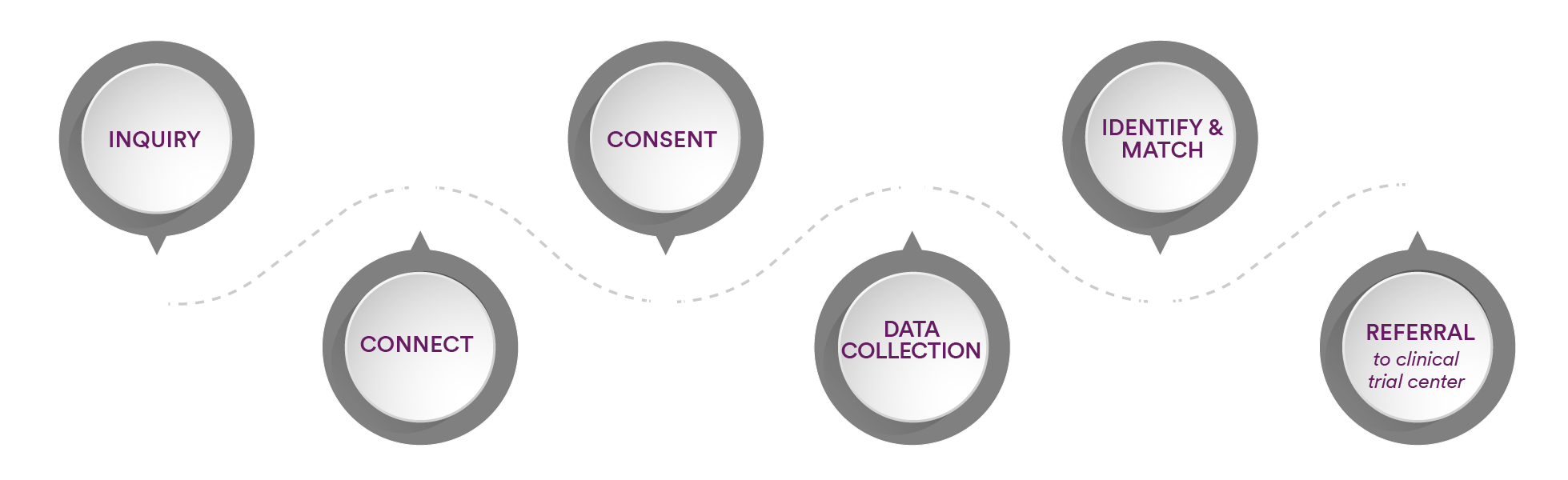
When contacting SareptAlly, individuals will be connected to a Patient Navigator. The SareptAlly Patient Navigator will serve as a resource for identifying potential clinical trial options or local treatment access. The Patient Navigator is a medically trained professional whose focus is to understand the unique circumstances of individuals and provide discrete, personalized options. For clinical trials, here’s how the process works:
- Make an appointment with a Patient Navigator using the scheduler link.
- Speak with the Patient Navigator
- Provide consent for services
- Share personal health information to help determine which Sarepta clinical trial options might be available for consideration
- Patient Navigator will discuss potential clinical trial options and locations where those clinical trials are conducted
- Patient Navigator will assist in connecting the patient to the clinical trial center.
SareptAlly Clinical Trial Identification FAQs
-
Does participation in SareptAlly guarantee a spot in a clinical trial?
The SareptAlly program is designed to help match individuals to physicians participating in Sarepta-sponsored clinical trials. The clinical trial physician will make the determination as to who is enrolled in a clinical study independent of Sarepta. Factors in their decision include:
- Clinical trial eligibility criteria such as age, diagnosis, or current or prior therapies
- The number of participants planned for a particular study.
-
What type of data are required for SareptAlly?
To participate in SareptAlly, some personal information is required, including name, contact information (phone number, email address), and country of residence. These data will allow the Patient Navigator to maintain contact and provide updates about potential clinical trials.
To help the Navigator better assist in identifying potential clinical trials, additional information, such as demographics and medical history, may be requested.
These data include but may not be limited to sex, age, weight, ambulation, mobility, or ability to perform certain functions. Depending on the clinical trial, the Patient Navigator may ask about certain diagnosis and genetic test results. Participants in SareptAlly may wish to provide the name of the treating physician to allow direct interaction with the Patient Navigator.
Requested information may be provided verbally or by sharing medical records. SareptAlly is committed to maintain confidentiality and will obtain consent before collecting any information. The participant in SareptAlly can make the determination whether or not to provide requested information.
-
What if there are no clinical trial options?
If there are no clinical trial options available, the Patient Navigator will discuss if local treatment access options are a possibility. If no local treatment option is available, the Patient Navigator will offer to connect at scheduled intervals such as every 3-6 months, to learn if new clinical trial options are available. Patients, family members, or physicians can call the Patient Navigator at any time.
-
Will the Patient Navigator follow up if new options become available?
The Patient Navigator is updated when new Sarepta-sponsored clinical trials are available and may contact individuals with new opportunities based on eligibility and location to clinical trial centers.
-
What if I have other questions outside of clinical trial enrollment and local treatment options
If you should have questions regarding other Sarepta-related topics, you may contact our Patient Affairs group at [email protected]
To learn more about ongoing clinical trials at Sarepta click here
What to Expect When Connecting with SareptAlly: Treatment Access Process
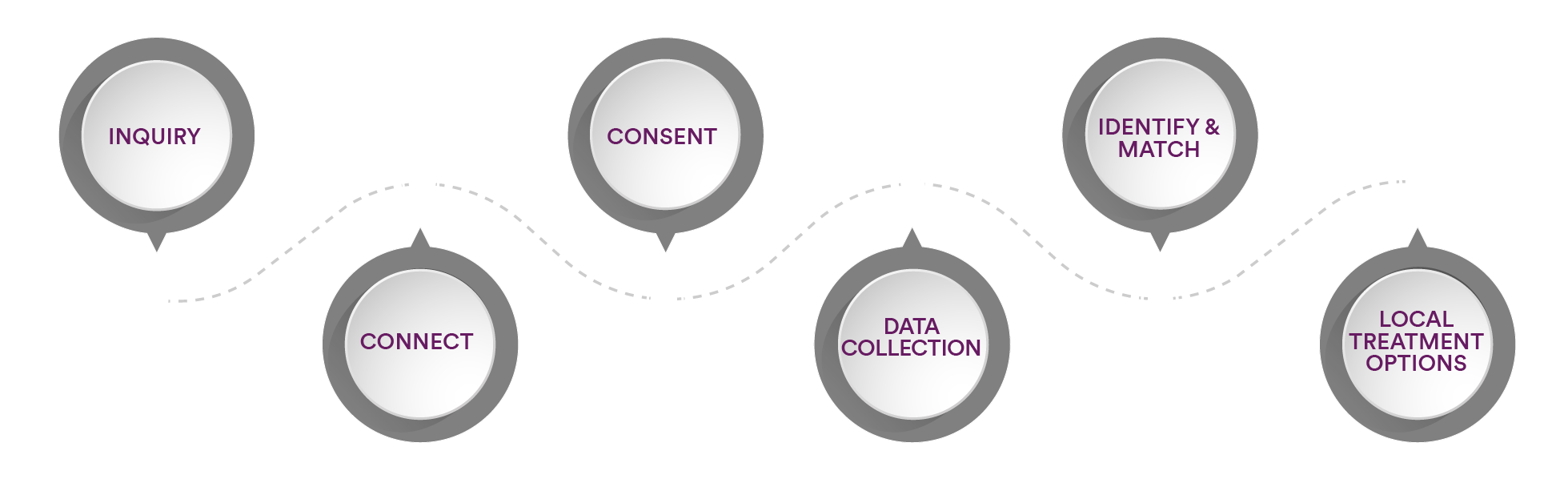
When contacting SareptAlly, the Patient Navigator may identify local treatment access. The overall process for engaging with SareptAlly remains the same. Here’s how it works:
- Make an appointment with a Patient Navigator using the scheduler link.
- Speak with the Patient Navigator
- Provide consent for services
- Share personal health information to help determine which options might be available for consideration
- If a potential treatment option is available, the Patient Navigator can review local country requirements and support the physician’s application and review process for that country.
SareptAlly Local Treatment Options FAQs
-
How can I find out what potential treatment options are available near where I live?
Once the intake appointment is scheduled, a Patient Navigator will work with patients, families, and their medical team to determine treatment eligibility. If eligibility criteria are met, the Patient Navigator will discuss how to pursue treatment access. The patient may have access to clinical trials, local distributors who provide treatment access, or early access to therapy options depending on the country where you live. Your Patient Navigator will review each option and work to identify appropriate next steps.
-
Are there other potential ways to access treatment in my country aside from a clinical trial?
Many countries offer early access or managed access programs to support access to unapproved medication. An early access program is a mechanism through which physicians can prescribe, within their professional discretion, an investigational treatment for patients. Use of an unapproved, investigational treatment is based on physicians’ informed clinical assessment to determine whether there is a special need for the unapproved treatment because (a) there is no approved treatment available or (b) available treatments are not suitable for that patient. To learn more, schedule an appointment with one of our Patient Navigators who can explain your options related to managed access programs in your country.
-
Where is this program available?
SareptAlly is available to all patients, families and physicians, globally. For those seeking treatment in the United States, SareptAssist is the local program dedicated to supporting access to Sarepta-approved therapies in the U.S. For more information, please go to SareptAssist
Contact SareptAlly
Email:
