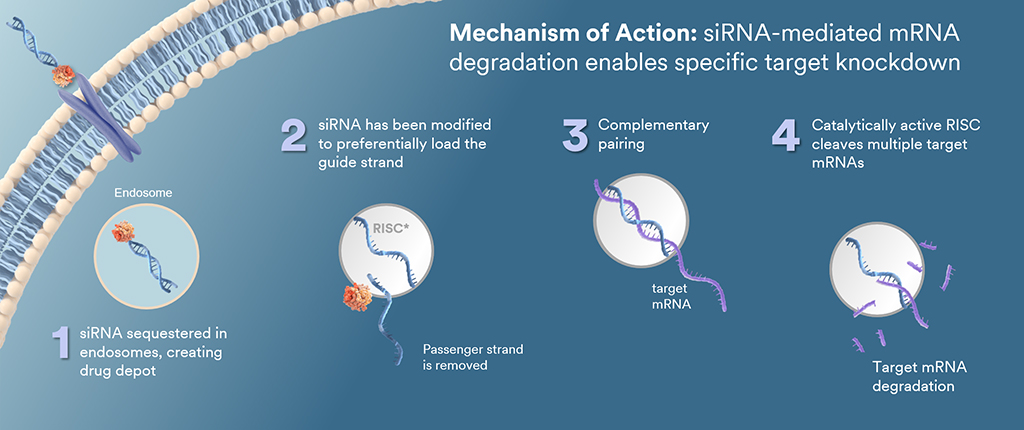
Below is a depiction of how TRiM proprietary technology works.
- The siRNA is taken up by tissue and into an endosome by a specific ligand.
- Modifications such as a stable phosphate on the guide strand have been made, aiming to create metabolic stability and efficient RISC loading.
- This next-generation approach aims to offer increased potency since a catalytically active RISC cleaves multiple targeted mRNAs.
The potential benefits of this technology include:
- Designing the chemistry to improve potency and specificity, thereby potentially mitigating off-target effects to achieve silencing of disease-causing mRNAs and proteins;
- By targeting mRNA directly these therapies have the potential to target any disease-causing protein, thereby increasing the number of potential drug targets; and
- Knocking down disease-causing mRNAs with the goal of reaching areas in the body that are difficult to penetrate.
Our lead programs leveraging this technology are being developed to treat facioscapulohumeral muscular dystrophy type 1 (FSHD1) and myotonic dystrophy type 1 (DM1) for which treatments do not currently exist.